Objectives
The objectives of this section are
to provide you, on the one hand, with a few more detailed examples how biofilms can be used in beneficial ways;
to give you, on the other hand, a few more detailed examples of some of the harmful impacts of biofilms.
Outcomes
Upon completion of this section, you will be able
to discuss more detailed examples of both the beneficial and harmful impacts of biofilms on our world;
to describe in an informal way how the biofilm mechanism works to achieve either a beneficial or harmful effect, based on the situation in which the biofilm appears.
How do biofilms impact our world?
Biofilms profoundly affect human health and industrial productivity. Their pervasive effects on human health, water quality, corrosion, and power generation efficiency cost the U.S. billions of dollars annually. Other related issues include deterioration of dental surfaces, contamination of surfaces in the food processing industry, and the deterioration of air quality in ventilation and air handling systems.
However, it is very important to point out that biofilms are an integral part of the natural environment and can also serve very beneficial purposes, such as in the treatment of drinking water, wastewater and detoxification of hazardous waste.
Both the beneficial and detrimental aspects of biofilms are summarized below.
BENEFICIAL BIOFILMS
In natural environments
In bodies of water both large and small, on land, underground, as well as on and within higher organisms, biofilms are an integral component of the natural environment. The report, "Global Environmental Change: Microbial Contributions, Microbial Solutions," points out: ". . .the basic chemistry of Earth's surface is determined by biological activity, especially that of the many trillions of microbes in soil and water. Microbes make up the majority of the living biomass on Earth and, as such, have major roles in the recycling of elements vital to life." Bacteria and other microorganisms have also been associated with the formation of many types of sedimentary rocks and minerals.
Bacteria are early colonizers of clean surfaces submerged in water. Scientists have been able to document the predictable sequential colonization of surfaces in ocean waters by series of organisms, beginning with microbial biofilms. Whether the surface is a boat hull at the surface or a new deep sea vent at the bottom of the ocean, microbes are available and capable of rapid community development on surfaces. Microbes have evolved along with other organisms, including human beings. While some bacteria produce effects that are detrimental to surrounding organisms or hosts, most bacteria are harmless or even beneficial. When it comes to bacteria, higher organisms are just another environment to colonize.
Water and wastewater treatment
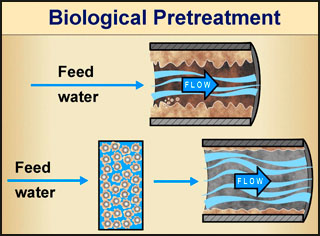
Engineers have taken advantage of natural biofilm environmental activity in developing water-treatment systems. Biofilms have been used successfully in water and wastewater treatment for over a century. English engineers developed the first sand filter treatment methods for both water and wastewater treatment in the 1860s. In these filtration systems the surfaces of the filter media act as a support for microbial attachment and growth, resulting in a biofilm adapted to using the organic matter found in that particular water. The end result of biological filtration is a conversion of organic carbon in the water into bacterial biomass. Ideally, this biomass is immobilized on the filter media and removed during the backwash cycle.
Drinking water and treated wastewater that have been subjected to microbial activity in a controlled manner in a treatment plant are more "biologically stable" and therefore less likely to contribute to microbial proliferation downstream in distribution system or receiving water. Biologically treated water typically has lower disinfectant demand and disinfection by-product formation potential than conventionally treated water if the source water is high in organic carbon. As drinking water utilities move to using ozone as a primary disinfectant and for taste/odor/color control, biological filters may be necessary to reduce the concentrations of biodegradable organic carbon entering the distribution system.
Remediation of contaminated soil and groundwater
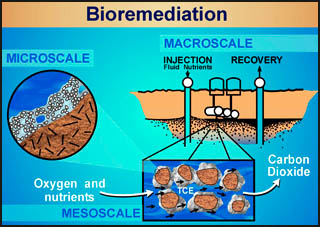
In soil, biofilm morphology can be highly variable, ranging from patchy discontinuous colonies to thick continuous films, depending on environmental conditions. When toxic organic contaminants (i.e. gasoline, fuel oil, chlorinated solvents) are accidentally released underground, the native soil bacterial population will, to the degree possible, adjust their ecological composition in order to use the organic contaminants as a food source. This process is commonly referred to as “bioremediation” and if successful, potentially has the ability to render initially toxic organic material into harmless by-products. Typical biofilm cell densities found in the vicinity of contaminated ground water sites vary from around 105 to 108 cells per gram of soil.
Bioremediation has emerged as a technology of choice for remediating groundwater and soil at many sites contaminated with hazardous wastes. Bioremediation results in 1) the reduction of both contaminant concentration and mass for many subsurface contaminants (e.g., petroleum hydrocarbons, chlorinated organics and nitroaromatics) and/or 2) a beneficial phase transfer or speciation change (e.g., for heavy metals and radionuclides). Subsurface bioremediation is controlled by abiotic geochemical and transport phenomena, including multiphase flow, convective mass transport, adsorption/desorption, and phase partitioning, as well as biotic processes, such as microbial biomass growth and contaminant metabolism
Microbial leaching

Photo courtesy of Kennecott Utah Copper, Salt Lake City, Utah. This image shows a vast complex of leaching fields in which ore of low copper content is sprayed with water. Bacteria such as Thiobacillus, attached to the surface of the ore particles oxidize the insoluble copper compounds to soluble copper sulfate from which the pure copper can easily be recovered.
Extraction of copper from ore deposits using acid solutions has been practiced for centuries, but the role of bacteria in metal dissolution was not verified until the 1940s. Today approximately 10–20% of copper mined in the U.S. is extracted by microbially assisted processing of low grade ores. There is also a considerable investment in extending microbial leaching to the recovery of other metals such as uranium, silver, gold, cobalt, and molybdenum. Most microbial leaching depends upon microbial oxidation of metal sulfides. Aqueous environments in association with spent mineral produce very harsh conditions of low pH, high metal concentrations and high temperatures, which select for a microbial flora with very specialized nutritional requirements.
Heap leaching is the most common process used to microbially extract copper and other minerals from spent ore. The process consists of organizing the spent ore fragments into a packed bed configuration which allows water to be trickled through. To initiate the process, acidified water (pH = 1.5 to 3.0) is sprayed over the porous ore bed. Acidophilic bacteria, such as Thiobacillus ferrooxidans, actively oxidize the soluble ferrous iron and attack the sulfide minerals, releasing the soluble cupric ion that can then be recovered from aqueous solution. This oxidation process is similar in concept to corrosion of metal surfaces.
Biological reaction and mass transfer rates presently limit the commercial application of microbial leaching, however substantial improvements in process design have been made in recent years and the method is viewed with promise by the mining industry.
DETRIMENTAL BIOFILM IMPACTS
Of course, not all biofilms work to our advantage. While sewage treatment plants and bioremediation are critical in efforts to keep out environment livable, each of these has a dark side. The nitrogen, phosphorous and organic waste from agriculture and inadequately treated municipal waste collected from half the continent by the Ohio, Missouri and Mississippi river systems end up in the Gulf of Mexico. These nutrients cause over enrichment of the waters of the Gulf, a process called eutrophication. This in turn causes explosive growth or blooms of algae. On dying, the decomposition of these same algae cause widespread oxygen depletion in the Gulf and the creation of anoxic “Dead Zones” where nothing that is dependent on oxygen can survive. The resulting death of fish and shellfish have had a devastating effect on the commercial fishing industry of the Gulf.
The damage done by the recent Deepwater Horizon oil spill and its affect on the Gulf fisheries must not be under estimated, but once the flow of oil is stopped, microorganisms will, in time, remove the oil and restore the health of the Gulf waters as they did following the Exxon Valdez spill in Prince William Sound Alaska. The dead zones are an ongoing problem caused largely by human farming practices and will continue until the over use of chemical fertilizers, and the release of municipal, industrial and animal waste is brought under control.
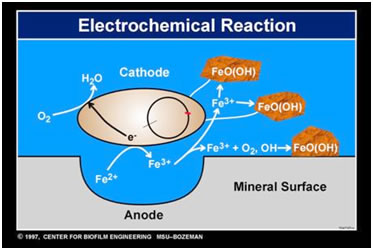
Just as there are bacteria such as Thiobacillus that can harvest copper from low grade ore, there are other bacteria that can act on metals causing what is called biocorrosion or MIC (microbially influenced corrosion). Perhaps chief among these are the SRBs or sulfate reducing bacteria. After attaching to the wall of a water distribution pipe, the tubing of a heat exchanger, or an oil delivery pipeline the bacteria form a biofilm and within the anaerobic zone of that community gain energy by pitting and corroding the surface of the metal. The result is billions of dollars of damage annually as pipes must be replaced or as they fail and cause further pollution.
The following stories illustrate three further examples in which biofilms may cause damage to humans or to the environment. Two of these are clearly anthropogenic the other not clearly so.

Legionnaire’s Disease
In microbiological terms, the bug is said to be fastidious, that is, in the laboratory it is pretty hard to grow. This is sort of surprising since in nature it grows in locations that one would expect to be very generous in terms of nutrients. Legionella pneumophila, is a bacterium widely spread in nature. It grows in soil, fresh water, hot tubs and interestingly as a luxuriant biofilm on the wooden slats or organic batting of the condensers of the air conditioning systems of buildings. L. pneumophila can survive under a great range of temperatures from 0 to 63 C (or 145 F) too hot to touch. Legionella does best, however, at just about human body temperature of 98.6F.
This organism escaped identification until an outbreak of a previously unidentified type of pneumonia was detected among the attendees at an American Legion convention in Philadelphia in July of 1976.

The convention that year was held in the venerable Belleview-Stratford Hotel and the hundreds of Legionnaires in attendence were mostly veterans of World War II and the Korean War.
These veterans, mostly in their mid 40s or older, were of an age where the effects of growing older are beginning to have an impact and many due to a number of predisposing causes (diabetes, asthma, emphysema) were medically compromised.
Beginning on the second day of the convention, a number of the delegates fell ill with an acute pneumonia-like disease and before the illness had run its course, over 200 Legionnaires had become ill and 30 had died of a disease, not previously recognized.
Eventually the causal organisms of this outbreak was identified by scientists at the Centers for Disease Control and it was named Legionella pneumophila after the first known victims of the disease and its primary site of attack, the lungs. The organism was found to be ubiquitous in distribution but had some rather unusual nutritional requirements (the amino acid cystine and Iron).
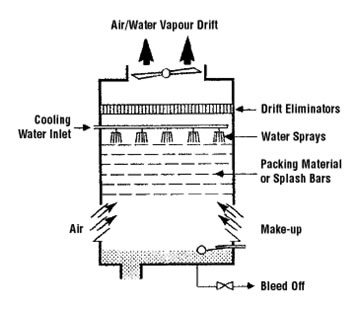
The source of infection was found to be the cooling towers of the air-conditioning units of the hotel in which the air circulated through the hotel was chilled by evaporational cooling. Water cascading over wooden baffles evaporated causing a cooling effect, which was used to cool the air for the hotel. Unfortunately, these baffles and the water reservoir below provided a near optimum environment for the luxuriant growth of Legionella. The vapor drift of evaporated water, carried with it large numbers of bacteria to the streets below where Legionnaires chatting with friends just outside the hotel were exposed. CDC data revealed that those veterans who had spent the most time in the street just outside the hotel were at greatest risk for Legionnaires Disease.
Although this was the first recognized case of this “new” disease, samples stored in freezers from previously unexplained cases of pneumonia revealed that it was not the first incidence. It was also not the last. Legionnaires Disease is now recognized as a rather common form of pneumonia and the organisms responsible for it has been cultured from hot-water tanks, shower heads, the faucets of sinks, hot tubs, indoor and outdoor water features like fountains and soil.
Fortunately, unlike the 1976 outbreak, there are now analytical methods for rapidly diagnosing Legionnaires Disease and antibiotics that are very effective in its treatment.
Black Band Coral Disease Adapted from NOAA’s Coral Health and Monitoring Program.
The dying begins with a white spot surrounded by a reddish or black band on an otherwise healthy coral formation. In an astonishingly short time the band grows outward in all directions producing a roughly circular region in which only the dead white coral skeleton remains. Growing at a rate of from a few mm to 1 cm a day this Black Band Disease can in the worst cases completely destroy a coral outcrop within months.
First described in the 1970s as a black band migrating across the surface a star and brain corals the disease is now know to have world wide distribution including outbreaks in the Caribbean and in the Florida Keys.
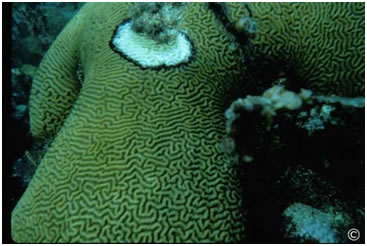
The pathogens responsible for this disease are in fact a consortium of bacteria consisting of photosynthetic cyanobacteria (e.g. Phormidium coallyticum), sulfide-oxidizing bacteria (Biggiatoa spp.) and sulfate reducing bacteria (Desulfovibrio spp.) plus an large array (perhaps as many as 500) other bacterial species not found in health coral tissue or in the adjacent water column. The black (or red band) is due to pigments (phycoerythrins) produced by the cyanobacterium.
The presumed lethal event, however, is the formation of an oxygen-deprived zone rich in hydrogen sulfide and produced by the combined activities of the sulfate reducing and the sulfide-oxidizing bacteria. Sulfate reducing bacteria growing deep with in the black band reduce sulfate from seawater producing large amounts of sulfide which is subsequently oxidized by Biggiatoa, aprocess that utilizes virtually all the available oxygen and produces toxic hydrogen sulfide. The coral animals or polyps die from the combined effects of oxygen deprivation and H2S poisoning as the biofilm band passes over them, leaving only their bleached skeletons behind.
Hot-tub Folliculitis
The pool and hot tub party had gone off with out a hitch. The 33 children who attended had a great time and could hardly stop talking about the fun they had had swimming and soaking for hours with their friends. But later that evening and over the next two days, 32 of the children began to develop small reddish lesions located on various regions of their bodies. Two of the children, a nine-year old girl and a four-year old boy, developed low grade fever (about 99F) and were admitted to the hospital for diagnosis and treatment.
Laboratory tests revealed an increased white blood cell count, an indication of infection, and both exhibited a reddened nodules of the skin (erythema) on their palms and soles of their feet. One, the girl, developed reddish pustules associated with hair follicules on her head and trunk. This observation led to the diagnosis of folliculitis1.
Bacteriological cultures taken from the pustules grew Pseudomonas aeruginosa, a ubiquitous bacterium that dwells in soil and water and can tolerate high temperatures of the sort encountered in hot tubs. P. aeruginosa was also grown from samples of water recovered from the hot tub and DNA analysis showed the strains from the tub and the children to be nearly identical.
Both the children were placed on oral ciprofloxacin and within a few days the red pustules had completely gone away.
Bacterial folliculitis is generally regarded as a relatively benign self-limiting infection, but it is the sort of thing that can ruin a long planned vacation.
Proper maintenance of pools and particularly hot tubs is critical in the prevention of this aggravating condition. Frequent cleaning and the maintenance of adequate chlorine or bromine levels is necessary. Attention to these details is probably even more important in hot tubs than in swimming pools due to the smaller volume and increased bacterial contamination load.
How to avoid Hot Tub Folliculitis
Check out the hot tub, if it is slimy or sticky this is a sign of the presence of a biofilm and the failure of proper spa maintanence, it might be best to try the pool.
If the hot tub has a funky odor (other than the disinfectant) avoid it.
Shower with soap and hot water before entering and after leaving the Hot Tub.
Wash out your bathing suit in hot soapy water after using the hot tub. Dry it thoroughly between uses.
Remember, diabetes, skin infections or open sores increase your risk of hot tub folliculitis. The rash is typically more severe in areas where your swimsuit held the chlorinated water against your body.
Four Questions to Ask Your Hot Tub Operator
- What was the most recent health inspection score for the hot tub?
- Are disinfectant and pH levels checked at least twice per day?
- Are disinfectant and pH levels checked more often when the hot tub is being used by a lot of people?
- Are the following maintenance activities performed regularly:
- Removal of the slime or biofilm layer by scrubbing and cleaning?
- Replacement of the hot tub water filter according to manufacturer’s recommendations?
- Replacement of hot tub water?
From the CDC recommendations on preventing Hot Tub Rash. http://www.cdc.gov/healthywater/swimming/rwi/illnesses/hot-tub-rash.html
Reference
1 Yue Yu, Amy S. Cheng, Lawrence Wang, W. Michael Dunne and Susan J. Bayliss, 2007. Hot Tub folliculitis or hot Hand-foot syndrome caused by Pseudomonas aeruginosa, Journal of the American Academy of Dermatology, 57: 596-600.


